HAL Robotic Remedial Device Helps Disabled Persons From Diseases Like MD Re-learn How To Walk

Japan’s Cyberdyne, Inc. has applied for marketing authorization of their HAL for Medical Use (Lower Limb Type) robot exoskeleton as a new medical device for orphan neural-muscular diseases, such as muscular dystrophy, amyotrophic lateral sclerosis (ALS) and others under Japan’s Pharmaceutical and Medical Device Law (PMDL). Under the law, new medical devices are required to have different mechanisms, directions for use, and efficacies or effect from those of previously existing medical devices. Applications for new medical devices require attachment of clinical test results.
Cyberdyne claims that HAL (Hybrid Assistive Limb) is the world’s first cyborg-type robot for enabling, supporting enhancing and improving a wearer’s motor function ability and performance, describing HAL as leading to a fusion of man, machine and information.
HAL Therapy is the medical service with Robot Suit HAL to provide medical treatments for functional improvement of patients with cerebral, nervous and muscle disorders including spinal cord injury and cerebral embolism.
 HAL assists a physically challenged person to move and enables him or her to exert greater motor energy. HAL is also intended to accelerate motor learning of cerebral nerves. The international patent relevant to HAL has been accredited as a Notable Invention by the World Intellectual Property Organization (WIPO), an agency of the United Nations was created in 1967 as a global forum for intellectual property services, policy, information and cooperation.
HAL assists a physically challenged person to move and enables him or her to exert greater motor energy. HAL is also intended to accelerate motor learning of cerebral nerves. The international patent relevant to HAL has been accredited as a Notable Invention by the World Intellectual Property Organization (WIPO), an agency of the United Nations was created in 1967 as a global forum for intellectual property services, policy, information and cooperation.
HAL Therapy is the medical service with Robot Suit HAL to provide medical treatments for functional improvement of patients with cerebral, nervous and muscle disorders including spinal cord injury and cerebral embolism.
Major causes of lower limb disabilities are disorders of the cerebral and nervous muscular system. In those cases, the brain cannot use ordinary neural pathways and cannot order the neuromuscular system move the legs.
Human body movement does not involve only moving muscles. When a person intends to move his or her body, nerve signals are transmitted from the brain to muscles via motor neurons and then to the musculoskeletal system location such as a joint which executes the movement. At that time, subtle bio-electric signals leak out onto the skin surface as bio-electric signals (BES), reflecting his or her intention to move. HAL for Medical Use – Lower Limb Model reads the BES and controls a power unit that is able to assist the joint’s movement in harmony with the person, accordingly compensating lack of muscle power of the lower limbs, and assists him or her in walking, standing-up and sitting-down with his or her own legs.
HAL for Medical Use – Lower Limb Model moves the wearer’s legs in accordance with the wearer’s intention: “I want to walk,” or “I want to stand up,” and enables timely feedback of the feelings: “I could walk,” or “I could stand up.” Consequently, this accelerates learning by the brain of how to move the legs again.
The brain signals how the body is to move, and When HAL assists the motions of walking, the user derives the feeling: “I can walk!” which is fed back to the brain. Through this process, the brain is able to learn how to emit the necessary signals for walking gradually, which can be the important first step toward the physically challenged person learning to walk again without being assisted by HAL.
There is a hypothesis that when a patient uses HAL to provide motion assistance in line with BES as a reflection of motion intention, interactive Bio-Feedback [hereinafter referred to as iBF] is created among HAL, the cerebral/nervous system and the muscular system via or outside the human body. By iBF, functions of patients with cerebral/nervous system and the muscular system disorders will hypothetically be reduced.
Based on this iBF hypothesis, via clinical application, the theoretical potential for functional improvement in human walking is being confirmed. Described another way, not only is motion initiated by the transmission of the spontaneous command signal “I want to move!” from the human brain to the musculoskeletal system via the spinal cord and peripheral nerves, but also feedback of the actual feeling “I could move!” to the brain are the important keys to acceleration of functional improvement.
Treatments for functional improvements and functional regeneration are kinds of new robot therapies, which aim at improvement of motor functions using HAL based on the above-described iBF hypothesis.
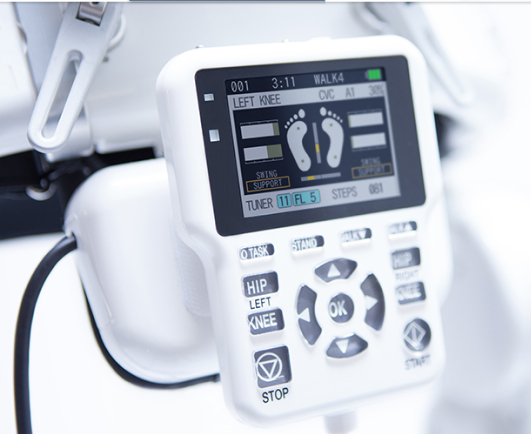
With the HAL’s freely detachable controller, the operator can manipulate all operations, such as start/stop of assistance, alteration of settings, and confirmation of motion status, closely monitoring the wearer’s movements.
HAL for Medical Use Lower Limb Model can be tailored to the individual wearer’s leg lengths, hip widths, and foot sizes across a wide range of seamless adjustment. This tailor-made accommodation of individual wearers makes training with HAL for Medical Use – Lower Limb Model more effective.
HAL technology can be applied to various fields, such as motion assistance for the disabled, assistance for heavy work in factories and other industrial settings and assistance for rescue activities at accident or disaster sites.
HAL for Medical Use – Lower Limb Model is approved in the EU as a medical device for patients with Musculoskeletal Ambulation Disability Symptom Complex (MADS) who suffer from spinal cord injuries, traumatic brain injuries, cerebrovascular diseases, diseases of the brain and neuromuscular system, and suchlike.
Cyberdyne Inc. and its German business partner, Berufsgenossenschaft Rohstoffe und chemissche Industrie (Professional Association of Raw Materials and Chemical Industry (BG RCI), have jointly established Cyberdyne Care Robotics GmbH in Bochum, Nordrhein-Westfalen (NRW) Germany, aimed at providing treatment for functional improvement of patients with cerebral, nervous and muscular system disorders including spinal injury and cerebral embolism. This marks a milestone passed in advancing the Japanese robotic remedial device HAL toward becoming an internationally recognized medical technology and therapeutic measure. The HAL device has been accredited Certification of conformity to the EC Medical Device Directive (MDD) marking first time accreditation has been granted a robotic remedial device by the ECs largest notified body. This enables CYBERDYNE Inc. to indicate CE Marking for Medical Device, which requires periodic examination, on their Robot Suit HAL product, for distributing and marketing it as a medical device in the whole EU region.
[adrotate group=”3″]
In view of various insurance applications in the whole EU region, dissemination of treatment with HAL is expected. The company notes that Deutsche Gesetzliche Unfallversicherung [German Statutory Accident Insurance, (DGUV) decided to apply workman’s accident compensation insurance to the treatment for functional improvement utilizing Robot Suit HAL. The fee for medical treatment with HAL, of which one time fee is EU500 and number of times is up to 60 plus additional treatment, is fully covered by the compensation insurance.
CYBERDYNE Inc. was established in June, 2004 by Dr. Yoshiyuki Sankai of the University of Tsukuba, Japan, in order to materialize his idea for the HAL Robot exoskeleton in the field, of medicine, caregiving, welfare, labor, heavy work, entertainment and so forth. HAL was developed with the technologies created in Sankai Laboratory of Tsukuba University as an application of Cybernics a new integrated technology of human, robot and information systems and scientific interdisciplinary research domain centered on cybernetics, mechatronics, and informatics, and integrates neuroscience, robotics, systems engineering, information technology, kansei engineering, ergonomics, physiology, social science, law, ethics, management, and economics. Cyberdyne, making full use of Cybernics, will tackle and solve social issues via R&D and implementation of innovative technology coexisting with people, and will create a New Industry based on these solutions. As a result, new pioneers with experience in this new design process will emerge from using leading-edge innovative technology. To achieve this vision, Cyberdyne proposes “Techno-Peer Support, mutually supported by both humans and technology.”
Sources:
Cyberdyne Inc.
Image Credits
Cyberdyne Inc.